


Source Borley Textbook



Source Borley Textbook


Source Borley Textbook
The term “juvenile periodontitis” was introduced by Chaput and colleagues in 1967 and by Butler in 1969. In 1971, Baer definedit as “a disease of the periodontium occurring in an otherwise healthy adolescent which is characterized by a rapid loss of alveolar bone about more than one tooth of the permanent dentition.
• Usually affecting persons under 30 years of age (however, may be older).
• Generalized proximal attachment loss affecting at least three teeth other than first molars and incisors.
• Pronounced episodic nature of periodontal destruction. • Poor serum antibody response to infecting agents.
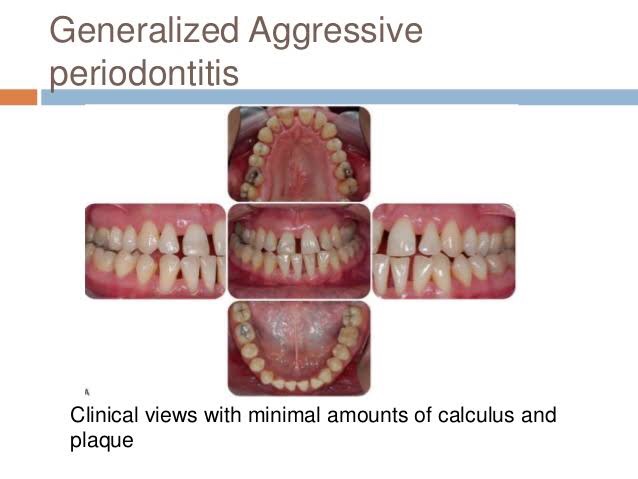
Clinical Characteristics
GAP usually affects individuals under age 30, but older patients also may be affected. In contrast to LAP, evidence suggests that individuals affected with GAP produce a poor antibody response to the pathogens present.
Clinically, GAP is characterized by “gen- eralized interproximal attachment loss affecting at least three per- manent teeth other than first molars and incisors.”The destruction appears to occur episodically, with periods of advanced destruction followed by stages of quiescence of variable length (weeks to months or years).
P. gingivalis, A. actinomycetemcomitans, and Tannerella forsythia (formerly Bacteroides forsythus) frequently are detected in the plaque that is present.
Two gingival tissue responses can be found in cases of GAP. One is a severe, acutely inflamed tissue, often proliferating, ulcerated, and fiery red. One is a severe, acutely inflamed tissue, often proliferating, ulcerated, and fiery red.
Bleeding may occur spontaneously or with slight stimulation. Suppuration may be an important feature. This tissue response is believed to occur in the destructive stage, in which attachment and bone are actively lost.
Some patients with GAP may have systemic manifestations, such as weight loss, mental depression, and general malaise.
Radiographic Findings
No definite pattern of distribution occurs, The radiographic picture in GAP can range from severe bone loss associated with the minimal number of teeth to advanced bone loss affecting the majority of teeth in the dentition.

Prevalence and Distribution by Age
and Gender
A US national survey of adolescents ages 14 to 17 reported that 0.13% had GAP. In addition, blacks were at much higher risk than whites for all forms of aggressive periodontitis, and male teenagers were more likely to have GAP than female adolescents.
Treatment of aggressive periodontitis
• Successful treatment of EOP depends on early diagnosis use of antibiotics against the infecting micro organisms and provision of an infection free environment for healing
• EOP both localized and generalized types includes surgery and use of tetracyclines (Lindhe 1982, Christersson and Zambon 1993)
• T/T of GAP – often less predictable – alternative antibiotics directed to specific pathogenic flora require
– Multidisplinary approach combines clinical labroartory evaluation with conventional periodontal therapeutic methods for diagnosis and t/t of GAP
Antibiotic Therapy for Aggressive Periodontitis
| Associated Microflora | Antibiotic of Choice |
| Gram-positive organisms | Amoxicillin–clavulanate potassium (Augmentin)12,72 |
| Gram-negative organisms | Clindamycin22,23,68,72 |
| Nonoral gram-negative, facultative rods | Ciprofloxacin41 |
| Pseudomonads, staphylococci | |
| Black-pigmented bacteria and spirochetes | Metronidazole22,65 |
| Prevotella intermedia, Porphyromonas gingivalis | Tetracycline55 |
| Actinobacillus actinomycetemcomitans | Metronidazole-amoxicillin22,65 Metronidazole-ciprofloxacin Tetracycline53 |
| P. gingivalis | Azithromycin54 |
REFERENCE- Caranza textbook of periodontology 11edition and Mc donald 9thed
The term “juvenile periodontitis” was introduced by Chaput and colleagues in 1967 and by Butler in 1969. In 1971, Baer defined it as “a disease of the periodontium occurring in an otherwise healthy adolescent which is characterized by a rapid loss of alveolar bone about more than one tooth of the permanent dentition.
According to Hart et al. diagnosis of localized early-onset periodontitis is based on
• attachment loss of > or equal 4 mm on at least two permanent first molars
and incisors (one of which must be a first permanent molar)
Not more than two other permanent teeth, which are not first permanent molars or incisors, should be affected
• Bone loss around primary teeth can be early finding in LAP

Clinical Characteristics
The lack of clinical inflammation despite the presence of deep periodontal pockets and advanced bone loss .
The amount of plaque on the affected teeth is minimal, which seems inconsistent with the amount of periodontal destruction present.The plaque that is present forms a thin biofilm on the teeth and rarely mineralizes to form calculus.
The rate of bone loss is about three to four times faster than in chronic periodontitis.
Other clinical features of LAP may include (1) distolabial migration of the maxillary incisors with concomitant diastema formation, (2) increasing mobility of the maxillary and mandibular incisors and first molars, (3) sensitivity of denuded root surfaces to thermal and tactile stimuli, and (4) deep, dull, radiating pain during mastication, probably caused by irritation of the supporting structures by mobile teeth and impacted food.
Radiographic Findings
Vertical loss of alveolar bone around the first molars and incisors, beginning around puberty in otherwise healthy teenagers, is a classic diagnostic sign of LAP. Radiographic findings may include an “arcshaped loss of alveolar bone extending from the distal surface of the second premolar to the mesial surface of the second molar”

Prevalence and Distribution by Age
and Gender
LAP affects both males and females and is seen most frequently in the period between puberty and 20 years of age.
RISK FACTORS FOR AGGRESSIVE PERIODONTITIS
Microbiologic Factors
A. actinomycetemcomitans, Capnocytophagaspp., Eikenella corrodens, Prevotella intermedia, and Campylobacter rectus), A. actinomycetemcomitans has been implicated as the primary pathogen associated with LAP.
Immunologic Factors
Some immune defects have been implicated in the pathogenesis of aggressive periodontitis. The human leukocyte antigens (HLAs), which regulate immune responses, have been evaluated as candidate markers for aggressive periodontitis.
Genetic Factors
Familial pattern of alveolar bone loss and have implicated genetic factors in aggressive periodontitis.
Environmental Factors
The amount and duration of smoking are important variables that can influence the extent of destruction seen in young adults.46 Patients with GAP who smoke have more affected teeth and more loss of clinical attachment than nonsmoking patients with GAP.16 However, smoking may not have the same impact on attachment levels in younger patients with LAP.
Treatment
In ps with LAP,
Aa organisms penetrate into crevicular epithelium T/T with antibiotic alone such as 2 week course of doxycycline reduce Aa organisms
Surgical removal of infected crevicular epithelium and debridement of root surface during surgery while the patient is on a 14 day course of doxycycline hyclate (1gm per day) (Mandell and Sockaransky 1988, Saxen et al 1990).
Microdentex manufacturers the DMDx(Microdentex, FORT MYERS, Florida) test, a DNA test kit for establisting the risk of aggressive periodontits and confirms whether the child has responded favorably to the use of antimicrobial therapy Retesting after 4-6 weeks after the completion of antibiotic therapy determines the pts response to t/t
• Rams and collagues described keyes technique for treating LAP Scaling and root planning of all teeth , with irrigation to probing depth of saturated inorganic salt solutions and 1% chloramine T.((sodium para-toluene sulfonchloramide) In addition recommended administration of systemic tetracycline (18 per day) for 14 days This dose appropriate for pts 12 yrs of age and older
Home T/T Daily application of paste of sodium bicarbonate and 3% hydrogen peroxide and inorganic salt irrigation.
| Associated Microflora | Antibiotic of Choice |
| Gram-positive organisms | Amoxicillin–clavulanate potassium (Augmentin)12,72 |
| Gram-negative organisms | Clindamycin22,23,68,72 |
| Nonoral gram-negative, facultative rods | Ciprofloxacin41 |
| Pseudomonads, staphylococci | |
| Black-pigmented bacteria and spirochetes | Metronidazole22,65 |
| Prevotella intermedia, Porphyromonas gingivalis | Tetracycline55 |
| Actinobacillus actinomycetemcomitans | Metronidazole-amoxicillin22,65 Metronidazole-ciprofloxacin Tetracycline53 |
| P. gingivalis | Azithromycin54 |
REFERENCE-
Caranza textbook of periodontology 11edition and Mc Donalds 9thedition
Pathologic changes in gingivitis are associated with the presence of oral microorganisms attached to the tooth and perhaps in or near the gingival sulcus.
STAGE I GINGIVITIS: THE INITIAL LESION
The first manifestations of gingival inflammation are vascular changes consisting of dilated capillaries and increased blood flow. These initial inflammatory changes occur in response to microbial activation of resident leukocytes and the subsequent stimulation of endothelial cells. Clinically, this initial response of the gingiva to bacterial plaque is not apparent.
Changes can also be detected in the junctional epithelium and perivascular connective tissue at this early stage. For example, the perivascular connective tissue matrix becomes altered, and there is exudation and deposition of fibrin in the affected area. Also, lymphocytes soon begin to accumulate. The increase in the migration of leukocytes and their accumulation within the gingival sulcus may be correlated with an increase in the flow of gingival fluid into the sulcus.
The character and intensity of the host response determine whether this initial lesion resolves rapidly, with the restoration of the tissue to a normal state, or evolves into a chronic inflammatory lesion. If the latter occurs, an infiltrate of macrophages and lymphoid cells appears within a few days.
STAGE II GINGIVITIS: THE EARLY LESION
The early lesion evolves from the initial lesion within about 1 week after the beginning of plaque accumulation.Clinically, the early lesion may appear as early gingivitis, and it overlaps with and evolves from the initial lesion with no clear-cut dividing line. As time goes on, clinical signs of erythema may appear, mainly because of the proliferation of capillaries and increased formation of capillary loops between rete pegs or ridges . Bleeding on probing may also be evident.1 Gingival fluid flow and the numbers of transmigrating leukocytes reach their maximum between 6 and 12 days after the onset of clinical gingivitis.
The amount of collagen destruction increases 70% of the collagen is destroyed around the cellular infiltrate. The main fiber groups affected appear to be the circular and dentogingival fiber assemblies. Alterations in blood vessel morphologic features and vascular bed patterns have also been described.
PMNs that have left the blood vessels in response to chemo- tactic stimuli from plaque components travel to the epithelium,
cross the basement lamina, and are found in the epithelium, emerg- ing in the pocket area. PMNs are attracted to bacteria and engulf them in the process of phagocytosis . PMNs release their lysosomes in association with the ingetion of bacteria.Fibroblasts show cytotoxic alterations, with a decreased capacity for collagen.
Meanwhile, on the opposite side of molecular events, collagen degradation is related to matrix metalloproteins (MMPs). Different MMPs are responsible for extracellular matrix remodeling within 7 days of inflammation, which is directly related to MMP-2 and MMP-9 production and activation.
STAGE III GINGIVITIS: THE ESTABLISHED LESION
Over time, the established lesion evolves, characterized by a predominance of plasma cells and B lymphocytes and probably in conjuncation with the creation of a small gingival pocket lined with a pocket epithelium.The B cells found in the established lesion are pre- dominantly of the immunoglobulin G1 (IgG1) and G3 (IgG3) subclasses.
In chronic gingivitis, which occurs 2 to 3 weeks after the beginning of plaque accumulation, the blood vessels become engorged and congested, venous return is impaired, and the blood flow becomes sluggish .The result is localized gingival anoxemia, which superimposes a somewhat bluish hue on the reddenedgingiva.18 Extravasation of erythrocytes into the connective tissue and breakdown of hemoglobin into its component pigments can also deepen the color of the chronically inflamed gingiva. The established lesion can be described as moderately to severely inflamed gingiva.
An inverse relationship appears to exist between the number of intact collagen bundles and the number of inflammatory cells.Collagenolytic activity is increased in inflamed gingival tissue17 by the enzyme collagenase. Collagenase is normally present in gingival tissues5 and is produced by some oral bacteria and by PMNs.
Enzyme histochemistry studies have shown that chronically inflamed gingivae have elevated levels of acid and alkaline phos- phatase, β-glucuronidase, β-glucosidase, β-galactosidase, esterases, aminopeptidase, and cytochrome oxidase. Neutral mucopolysaccharide levels are decreased, presumably as a result of degradation of the ground substance.
Established lesions of two types appear to exist; some remain stable and do not progress for months or years and others seem to become more active and to convert to progressively destructive lesions. Also, the established lesions appear to be reversible in that the sequence of events occurring in the tissues as a result of successful periodontal therapy seems to be essentially the reverse of the sequence of events observed as gingivitis develops. As the flora reverts from that characteristically associated with destructive lesions to that associated with periodontal health, the percentage of plasma cells decreases greatly, and the lymphocyte population increases proportionately.
STAGE IV GINGIVITIS: THE ADVANCED LESION
Extension of the lesion into alveolar bone characterizes a fourth stage known as the advanced lesion or phase of periodontal break- down.
Gingivitis will progress to periodontitis only in individuals who are susceptible. Patients who had sites with consistent bleeding had 70% more attachment loss than at sites that were not inflamed consistently (GI = 0). Teeth with noninflamed sites consistently had a 50-year survival rate of 99.5%, whereas teeth with consistently inflamed gingiva had a 63.4% survival rate over 50 years. Based on this longitudinal study on the natural history of periodontitis in a well-maintained male population, per- sistent gingivitis represents a risk factor for periodontal attachment loss and for tooth loss.
Reference- Caranza textbook of periodontology 11th edition
▪Can only be confirmed by determined by biopsy which is not always done.
▪Not always clear whether it is hyperplasia or hypertrophy.
▪Gingival enlargement is preferred terminology.
▪Vincent’s angina, Scurvy
▪Sodium Valproate, Nifedipine, Ciclosporin
▪Pregnancy, Oral contraceptive pill
▪Acute Myeloid leukaemia (M5) – may regress after treatment.
▪May be bleeding enlarged gums
▪Anaemia – ? AML
▪Known epilepsy
Management
▪Specialist review of medications as appropriate
▪Good dental care
Reference:Textbook of periodontology shantipriya Reddy


Source Carranza textbook


Source Carranza textbook


Source Carranza textbook



Source Carranza textbook